Posted August 13, 2012: by Bill Sardi
One of the dirty little secrets of modern medicine, and a reason why healthcare costs are so high, is that researchers and clinicians are trying to profiteer by inventing and delivering the highest cost medicine. Ten-cent cures are not to be found. Patentable, costly man-made medicines reign. Natural medicines are shunned or ignored.
Such is the case where British scientists claim they have discovered high iron levels in the colon/rectum raise the risk for cancer and say they are avidly searching for chelating (key-lay-ting) molecules that will mop up the iron and reduce the risk or even treat active colon/rectal cancer. They say they hope to start using these molecules in the coming years. But high-risk individuals with polyps in their colon or who have a history of bowel inflammation can reach for a cure that is already at hand.
A molecular cure already exists and is found in nature. It is the reason why whole-grain and high-fiber diets reduce the risk for colon/rectal cancer. A master iron-chelating molecule called IP6 (for inositol hexa-phosphate), found in the bran portion of grains and seeds, which is an available dietary supplement as a purified rice bran extract, has been demonstrated to inhibit colon/rectal cancer in numerous studies and inactivates the very same cancer-promoting gene (Wnt) shown in the newly published study in Britain. In one study IP6 decreased the uptake of iron in cells found in the colon by 50-65%.
This landmark study, published in journal Cell Reports, is stunning. While high-iron diets (particularly highly-absorbable iron from red meat) have consistently been shown to increase the risk for colon cancer, iron blood levels are not consistently correlated with this malignancy. Researchers show all the damaging effects of iron are local – in the colon and rectum.
The striking discovery is that even a high-iron diet will not induce tumors in these digestive tract tissues as long as a tumor-suppressing gene called Apc is functional. But researchers note that uncontrolled iron appears to increase mutation rates in the Apc gene that in turn inactivates its cancer-inhibiting action. Then two iron-accumulating genes (transferrin receptor 1 and divalent metal transporter 1) are switched on and iron pools in these tissues that results in the activation of a tumor-promoting gene (Wnt).
Researchers say: “chelation of this pool of iron may provide a platform for therapeutic intervention.” A class of chelators that inhibits the Wnt cancer-promoting gene has been identified.
But while there are expensive and problematic iron-chelating drugs, the safest and most economical is IP6. In 2001 researchers at the Food & Drug Administration declared IP6 to be one of only four non-mutagenic iron-chelating molecules. It would be the first truly preventive cancer therapy as no preventive measures are currently practiced beyond early detection.
Expensive iron chelators, most which require intravenous administration, are not appropriate for high-risk individuals who do not have active forms of colon/rectal cancer. However, IP6 does butt heads with the Food & Drug Administration’s requirement that a dietary supplement cannot cure, treat or prevent any disease without being declared a drug.
But all high-risk patients need do is head for a local health food store, they don’t need permission from the FDA. A 30-day supply of IP6 capsules sells for around $10 compared to the prescription oral chelator deferasirox that sells as a generic drug for around for $599.00/30 -500 mg tablets. For comparison the annual cost of generic deferasirox would be ~$7188/year compared to a 2-capsules per day regimen of IP6 around $60/year. Intravenous chelators cost thousands of dollars per year.
A long list of side effects are associated with deferasirox. These include but are not limited to: stomach pain, nausea, vomiting, diarrhea
Some side effects can be serious, such as: hearing loss, vision problems, hives, rash, blisters, itching, difficulty breathing or swallowing, swelling of the face, throat, tongue, lips, eyes, hands, feet, ankles, or lower legs, hoarseness
It’s estimated that 30 to 50 percent of Americans over 50 have or will develop adenomas, but only between 1 and 10 percent of these polyps will progress to cancer in 5 to 10 years. So it would be overtreatment to prescribe oral chelators for everyone who has diagnosed intestinal polyps.
A link between excess iron and colon cancer incidence has been repeatedly reported. Among 33studies, 75% of them support an association between excess iron and colon cancer risk.
The rationale to reduce the iron load is strong. Researchers already know that depletion of stored iron via blood donation or blood-letting decreases the risk for various cancers including colon cancer.
Researchers have been calling for human studies of IP6 since 2003. Another report about IP6 and cancer by this author was published in 2005.
A striking fact within this study is that patients at high-risk for colon cancer, those with polyps in their colon and rectum or who have inflammatory bowel disease, are often anemic due to binding of iron from chronic inflammation. The current practice among these anemic patients is to prescribe high dose oral iron tablets. The researchers in this study say this practice “is likely to both exacerbate their condition and elevate their risk for developing colon/rectal cancer.” (How many patients with anemia from chronic inflammatory bowel disease have met their early demise this way? This author can recall quite a few in his experience.) These researchers suggest supplemental iron be given intravenously and followed by iron chelation therapy. How many more patients will die of iron therapy before the word gets out about this? – © 2012 Knowledge of Health Inc. Not for posting at other websites
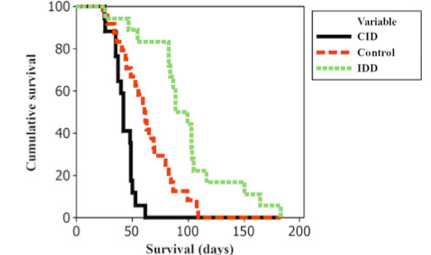
Survival of laboratory animals given high-carbonyl iron diet (black, CID),control diet (red), and iron-deficient diet (green- IDD)
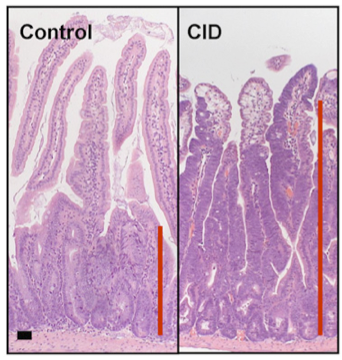
Appearance of colon cells on standard control diet (left) and high carbonyl iron diet (right)
|
Effect of intact or defective Apc gene and |
||
| Apc (adenomatous polyposis coli) Gene Is The Most Commonly Mutated Gene In Colon/Rectal Cancer |
Low Iron Diet |
|
| Intact (active) Apc gene Reduces cellular iron locally in the colon/rectum; protects Apc gene |
No cancer |
No cancer |
| Defective (mutated) Apc gene Increases cellular iron locally in colon/rectum; switches on Wnt tumor activator gene |
No cancer |
2-3 times increased risk |
| Defective Apc gene + intravenous (not local) iron chelator. Chelator must be taken orally and come in contact with cells in the colon and rectum |
No effect on tumor generation |
No effect on tumor generation |
| Source: Luminal Iron Levels Govern Intestinal Tumorigenesis after Apc Loss In Vivo, Cell Reports, 09 August 2012 | ||
Posted in Cancer, Dietary Supplements, Modern Medicine ; No Comments »
You must be logged in to post a comment.
11
17
52
95
14
24
237
6
56
43
10
116
15
66
105